HLA Typing and HLA Genes in Research: A Primer
Immunology is the study of the immune system, which includes both innate and adaptive immunity. This includes antibodies, specialized proteins that recognize foreign invaders such as bacteria or viruses; T cells, specialized white blood cells which are involved in cell-mediated immunity; and phagocytes (white blood cells) that engulf microbial invaders. Understanding these components of the immune system helps us to understand how our bodies resist disease and defend against external threats like pathogens. It also allows for better diagnosis, treatment, and prevention of various diseases by targeting specific parts of the immune system with drugs or vaccines. Finally, it can help us develop new strategies to combat infectious diseases as well as autoimmune disorders where a person’s own body mistakenly attacks itself.
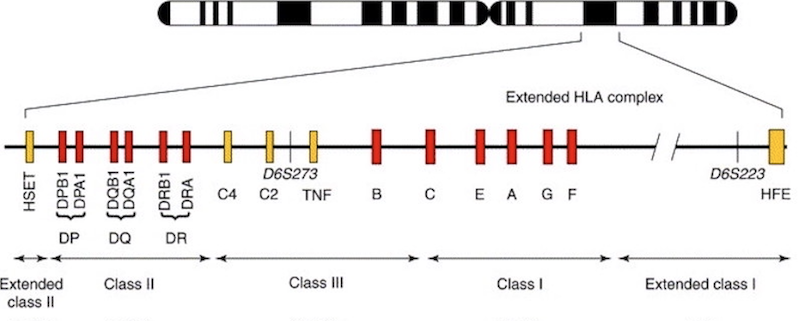
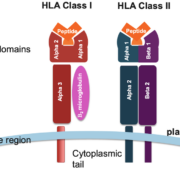
The human leukocyte antigen (HLA) genes are a group of major histocompatibility complex (MHC) molecules that play an essential role in the regulation of immune responses. The cluster of HLA genes is located on chromosome 6 and encodes for proteins found on the surface of cells, acting as receptors to foreign antigens. These proteins interact with other immune system components such as T-cells and B-cells, helping to identify invading pathogens and initiate an appropriate response by activating various defense mechanisms including inflammation and cytotoxicity. Additionally, HLA alleles can influence how well an individual responds to certain medications or treatments due to their structural similarity between inherited HLAs from one’s parents. This molecular recognition feature makes it possible for adaptive immunity responses like vaccination or organ transplantation surgeries to be successful.
The HLA genes also play an important role in determining tissue compatibility and transplantation success. When tissues or organs from one person are transferred to another, HLA plays an essential role in ensuring their acceptance by the new body. During successful transplants, donor cells must be recognized by recipient immune cells as “self” and accepted into the host organism without causing rejection. This process relies heavily on differences between donor and recipient HLA types; this ensures that only compatible organs can be successfully transplanted between individuals with different genetic backgrounds. The closer the match between the donor and recipient HLA type, the higher chance for a successful transplant outcome with minimal risk of complications like organ rejection or graft-versus-host disease (GVHD).
In addition to tissue compatibility, recent studies have identified certain specific HLA alleles associated with improved survival outcomes after heart transplantation, demonstrating the further importance of matching patients’ HLA type prior to any kind of organ transfer procedure.
Overall, due to its ability to predict cell recognition patterns across individuals with diverse genetic backgrounds, understanding how HLAs influence tissue compatibility has become an invaluable tool when it comes to increasing chances for a successful transplant outcome while minimizing risks associated with it
Variations in these genes may lead to different levels of response when exposed to pathogens or viruses as well as contribute to differences in other aspects such as drug efficacy. For example, if two individuals with the same genotype but different haplotypes were exposed to the same virus, one might show greater symptoms than the other because they possess different variants of HLA antigens that elicit different responses within their bodies’ immune systems. This is why it is important for medical researchers and practitioners alike to appreciate this variation among individuals when studying diseases such as HIV/AIDS or cancer, where particular types of HLA haplotypes are associated with more severe clinical presentations compared with others bearing similar alleles but differing haplotypes only slightly.
In addition, population-level variations in HLA genetics offer insight into how various groups react differently towards certain agents – like vaccines – yet share common characteristics overall; this helps guide public health strategies designed specifically for them based on their genetic profiles rather than merely by race or ethnicity alone. As more research accumulates regarding how individual variations affect disease risk and treatment effectiveness, so too will our understanding about how best to utilize this knowledge towards developing better treatments while simultaneously providing personalized care tailored according to each patient’s own unique genetic makeup.
In immunology research, HLA genes are studied to gain insight into why certain individuals may be more susceptible to certain infections or diseases compared with others. This knowledge can help researchers develop treatments and vaccines tailored to specific populations based on their genetic makeup. Additionally, understanding these complexes has enabled medical professionals to better predict transplant rejection rates by analyzing compatibility between donor and recipient’s HLA profiles prior to organ transfer. Therefore, it is clear that HLA research is critical for advancing modern medicine and improving public health outcomes worldwide.
Within infectious disease research, HLA genes encode for proteins that are essential for presenting foreign antigens to T cells, allowing them to recognize and eliminate invading pathogens such as viruses and bacteria. As a result, these molecules can be used to study infectious diseases by examining the association between particular infections and specific HLA alleles or haplotypes.
Studies have shown that certain HLA alleles may provide protection from acquiring some infections while increasing susceptibility to others. For instance, individuals with certain variants of HLA-B27 have been found to be more susceptible to developing ankylosing spondylitis after exposure to Klebsiella pneumoniae compared with people without this variant allele. Similarly, there is evidence suggesting that people who possess particular combinations of several different HLA allotypes may be at higher risk of infection with Epstein-Barr virus than those without them. By studying associations between various infectious agents and specific genetic loci encoding the various components of MHCs like those encoded by HLAs, researchers can further understand how susceptibility or resistance towards a disease might arise due to underlying genetics in humans which could help inform future strategies aimed at preventing or managing infectious diseases better in vulnerable populations.
To recap, HLA genes play a critical role in immunology research. They are responsible for the recognition and binding of antigens, which is essential for an effective immune response. HLA genes also regulate how T cells recognize and respond to foreign or self-antigens. In addition, they are involved in autoimmune diseases such as rheumatoid arthritis, type 1 diabetes, inflammatory bowel disease, and multiple sclerosis.
As the field of genetics continues to evolve scientists have been able to further understand the role of HLA genes in immunity; providing insight into potential treatments for many different types of illnesses. Recent advancements have allowed researchers to identify specific gene variants that may be associated with certain diseases or conditions including cancer susceptibility, organ transplant rejection, and autoimmunity disorders like lupus nephritis and psoriasis vulgaris.
There has also been an increased focus on developing personalized medical care based on one’s HLA profile – helping clinicians better diagnose illness by recognizing patterns between genetic profiles and various symptoms or risk factors for disease development/progression before treatment begins. Currently, there is ongoing research exploring methods to improve existing techniques used in haplotyping (the process of determining the exact sequence structure) so that individualized medicine can be applied more accurately across different patient populations.
HLA genes are integral components within immunology research due to their involvement with both antigen presentation processes as well as some forms of autoimmunity disorder development/expression; finding new ways better utilize this knowledge opens up numerous possibilities for improved healthcare outcomes through targeted therapies designed uniquely around individuals’ genomic information – something which we can expect further developments on over time!


 The Sequencing Center
The Sequencing Center

Leave a Reply
Want to join the discussion?Feel free to contribute!