What is the relationship between CAR-T and HLA Loci?
Chimeric Antigen Receptor T-cell (CAR-T) therapy represents a groundbreaking shift in our approach to treating malignancies, reshaping the landscape of cancer therapy. This revolutionary treatment involves engineering a patient’s own T cells to express a CAR that can specifically target tumor antigens, thus harnessing the body’s immune system to combat cancer. Intriguingly, the Human Leukocyte Antigen (HLA) locus and HLA typing bear significant implications for CAR-T therapy. This in-depth exploration will shed light on the interplay between CAR-T therapy, the HLA locus, and HLA typing, illuminating their potential impact on cancer treatment and personalized medicine.
CAR-T Therapy: An Overview
CAR-T therapy employs the body’s immune system to fight cancer, utilizing T cells engineered ex vivo to express a CAR, which can specifically recognize and bind to tumor antigens, independent of major histocompatibility complex (MHC) presentation. This strategy overcomes one of the key mechanisms by which tumor cells evade the immune system, namely downregulation or loss of MHC (HLA in humans) molecules. Thus, CAR-T cells can effectively target and eliminate tumor cells, even in the absence of MHC presentation of tumor antigens.
The Relationship between CAR-T Therapy and the HLA Locus
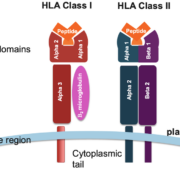
The HLA locus, which encodes the MHC molecules in humans, plays a central role in the immune response. Each individual’s HLA locus is highly polymorphic, leading to a diverse array of HLA types, which in turn results in variability in antigen presentation. This diversity is crucial for our ability to respond to a wide range of pathogens but also has significant implications for CAR-T therapy.
In conventional T-cell mediated immunity, T cells recognize antigens presented on MHC molecules by the antigen-presenting cells. The ability of a T cell to recognize a specific antigen-MHC complex is determined by the T cell receptor (TCR), and the specificity of this interaction is crucial for the immune response. However, tumor cells can evade this immune surveillance by downregulating or losing MHC molecules, rendering them invisible to T cells.
CAR-T therapy overcomes this challenge by bypassing the need for MHC presentation. The CAR expressed on the T cells can directly recognize and bind to the tumor antigens, irrespective of MHC presentation. This unique characteristic of CAR-T cells has significant advantages in combating tumors with low MHC expression, a common feature of many cancers.
HLA Typing and its Implications for CAR-T Therapy
While CAR-T therapy offers a promising strategy to tackle MHC loss in tumor cells, HLA typing still bears significant implications for this treatment. A major concern associated with CAR-T therapy is the potential for off-target effects, where CAR-T cells might attack healthy cells expressing the target antigen. Here, understanding the patient’s HLA type might help predict and manage potential off-target effects.
Moreover, HLA typing could have implications for the development of allogeneic CAR-T therapies, where CAR-T cells are derived from healthy donors rather than the patients themselves. In this scenario, matching the HLA types between the donor and the recipient could be crucial to minimize the risk of graft-versus-host disease and optimize the treatment’s efficacy.
Furthermore, although CAR-T therapy bypasses the need for MHC presentation, the interaction between the CAR-T cells and the patient’s immune system still involves HLA molecules. Understanding the patient’s HLA type could potentially help optimize the CAR-T cell design and the treatment strategy to enhance efficacy and minimize adverse effects.
Wrapping Up and Future Directions
CAR-T therapy represents a promising frontier in cancer treatment, harnessing the power of the immune system to combat malignancies. The HLA locus and HLA typing, despite the MHC-independent nature
of CAR-T cells, still have critical implications for this treatment strategy. These factors contribute to our understanding of the patient’s immune landscape, which can help predict potential off-target effects, inform the development of allogeneic CAR-T therapies, and potentially optimize CAR-T cell design.
While we have made significant strides in understanding and utilizing CAR-T therapy, much remains to be explored. How can we best utilize our knowledge of the HLA locus and HLA typing to optimize CAR-T therapies? Could HLA typing inform the choice of tumor antigen targets for CAR-T cells? Could it influence the design of the CAR itself or the choice of co-stimulatory domains to enhance CAR-T cell functionality and persistence?
As we delve into these questions, we find ourselves at the intersection of immunology, genetics, and oncology, combining our understanding of these fields to revolutionize cancer therapy. Future research in this area promises to unravel the complexities of the immune response against cancer, exploring the role of HLA in shaping this response and leveraging this knowledge to enhance the efficacy and safety of CAR-T therapies.
In the broader context of cancer treatment, CAR-T therapy is not alone in its potential for personalization based on the patient’s genetic makeup. The emergence of other forms of immunotherapy, such as checkpoint inhibitors and cancer vaccines, also provides opportunities for individualized treatment strategies informed by our understanding of the patient’s immune landscape, including their HLA type.
In conclusion, while CAR-T therapy can effectively target tumor cells regardless of MHC presentation, the HLA locus and HLA typing still have crucial roles to play in this treatment strategy. Whether it’s in predicting potential off-target effects, developing allogeneic CAR-T therapies, or potentially optimizing CAR-T cell design, our understanding of the HLA system can significantly influence the success of CAR-T therapy.
As we continue to explore these relationships and advance our understanding of the immune response against cancer, we can expect to see significant strides in personalized cancer treatment. Leveraging our knowledge of the HLA system and HLA typing can help maximize the potential of CAR-T therapy and other forms of immunotherapy, bringing us closer to the ultimate goal of providing optimal, personalized treatment for every cancer patient.



 The Sequencing Center
The Sequencing Center
Leave a Reply
Want to join the discussion?Feel free to contribute!